Three New Human Organs and Organ Systems Discovered!
Updated: Jan 2, 2020
Note: These new organs and organ systems are only know by a few scientists Worldwide and NOT currently taught in medical schools.
What is the Cause of Alzheimer’s, Parkinson’s and Huntington’s Disease? Is There a Potential Cure? Does the Discovery of Three New Human Organs and Organ Systems Help Us to Understand the Cause and Potential Cure?
Three new organs and organ systems discovered in the last two years include the Glymphatic System, the Interstitium System and the Meningeal Lymphatic System. These three systems, the Glymphatic System (or glymphatic clearance pathway, or paravascular system), the Interstitium System (the largest organ in all vertebrates) and finally the Meningeal System, functionally control the alkaline design of all body(pH) and act as an acidic waste clearance system for ALL vertebrates, of the central nervous system (CNS), the blood plasma, the interstitial fluids(ISF) and the intracellular fluids(ICF) within all animal and human body cells.[1]
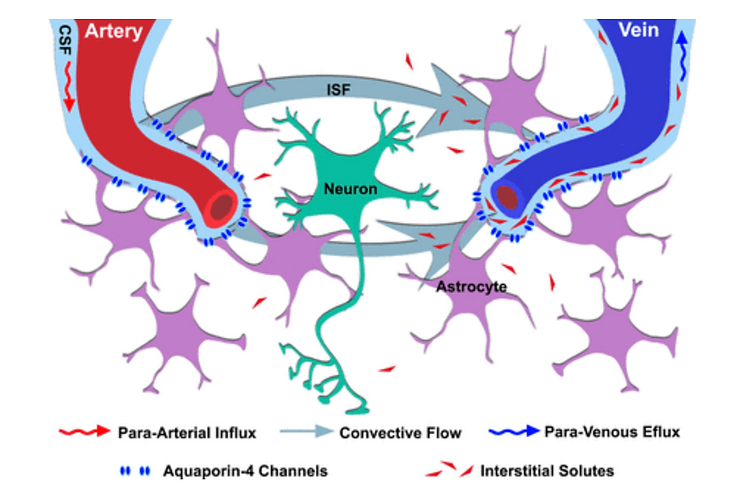
The pathway for the Glymphatic system consists of a para-arterial influx route for cerebrospinal fluid (CSF) to enter the brain parenchyma, coupled to a clearance mechanism for the removal of interstitial fluid (ISF) of the Interstitium and extracellular solutes from the interstitial compartments of the Interstitium in the brain and spinal cord. Exchange of solutes between CSF and ISF of the Interstitium is driven primarily by arterial pulsation[2] and regulated during deep sleep by the expansion and contraction of brain Interstitium compartment space. Clearance of soluble proteins, waste products, and excess extracellular fluid is accomplished through convective bulk flow of the ISF of the Interstitium, facilitated by astrocytic aquaporin 4 (AQP4) water channels.
The name “Glymphatic System” was coined by the Danish neuroscientist Maiken Nedergaard in recognition of its dependence upon glial cells and the similarity of its functions to those of the peripheral lymphatic system.[3]
While Glymphatic flow was initially believed to be the complete answer to the long-standing question of how the sensitive neural tissue of the CNS functions in the perceived absence of a lymphatic drainage pathway for extracellular proteins, excess fluid, and metabolic waste products, two subsequent articles by Louveau et al. from the University of Virginia School of Medicine and Aspelund et al. from the University of Helsinki reported independently the discovery that the dural sinuses and meningeal arteries are in fact lined with conventional lymphatic vessels, and that this long-elusive vasculature forms a connecting pathway to the Glymphatic System.[4][5] The Glymphatic System and the Interstitium work together to remove metabolic neurological waste during deep sleep via the newly discovered Meningeal Lymphatic System. [6][7][8]
When we do not have meaningful rest (8 hours), metabolic acids will build up in the interstitial fluids of the Interstitium compartments of the brain causing symptoms of light-headedness, muddle thinking, forgetfulness, dizziness, sleepiness, fatigue, diabetes mellitus, oxidative stress, dementia, Alzheimer’s, cranial pressure, headaches, including migraines, brain cancer, just to name a few. [9][10][11]
Pathologically, neurodegenerative diseases such as amyotrophic lateral sclerosis, Alzheimer’s disease, Parkinson’s disease and Huntington’s disease are all characterized by the progressive loss of neurons, cognitive decline, motor impairments, and sensory loss due to increased acidity within the Interstitium compartments..[12][13] Collectively these diseases fall within a broad category referred to as proteinopathies, due to the common assemblage of misfolded or aggregated protiens within the Intracellular fluids (ICF) and interstitial fluids of the Interstitium. According to the prevailing amyloid hypothesis of Alzheimer’s disease, the aggregation of amyloid-beta (a peptide normally produced in and cleared from the healthy young brain) into the I plaques drives the neuronal loss and brain atrophy that is the hallmark of Alzheimer’s dementia. Although the full extent of the Glymphatic sSystem’s involvement in Alzheimer’s disease and other neurodegenerative disorders is still under investigation, researchers have demonstrated through experiments with genetically modified mice that the proper function of the Glymphatic clearance system was necessary to remove soluble amyloid-beta from the brain Interstitium.[14] In mice that lack the AQP4 gene, amyloid-beta clearance of ISF is reduced by approximately 55 percent.
The Glymphatic system may also be impaired after acute brain injuries such as ischemic stroke, intracranial hemorrhage or subarachnoid hemorrhage. In 2014, a group of researchers from the French Institute of Health and Medical Research (INSERM) demonstrated by MRI that the Glymphatic System was impaired after subarachnoid hemorrhage, because of the presence of coagulated blood in the paravascular spaces.[15] Injection of tissue plasminogen activator (a fibrinolytic drug) in the CSF improved Glymphatic functioning and increased flow of ISF of the Interstitium and elimination via the Meningial Lymphatic System. In a parallel study, they also demonstrated that the Glymphatic System was impaired after ischemic stroke in the ischemic hemisphere, although the pathophysiological basis of this phenomenon remains unclear. Notably, recanalization of the occluded or blocked artery also reestablished the Glymphatic flow, the ISF flow and the elimination of acidic waste via the Meningeal Lymphatic System.

The schematic below is a representation of a connection between the Interstitium system and the Glymphatic system, responsible for collecting the interstitial fluids from within the CNS parenchyma to CSF, and our newly identified meningeal lymphatic vessels. This system is activated during deep sleep when the brain shrinks to push out metabolic acids into the Interstitium and then out via the meningeal lymphatic system. [16 -25]

Methodology for the Diagnosis of Interstitial Brain Disease (IBD) and All Other Associated Conditions
Investigation is tailored towards the symptoms and signs. A proper and detailed history looking for the occupational exposures, and for signs of conditions listed above is the first important part of the workup in patients with Interstitial brain disease (IBD) and its many symptoms. [26]

Testing the urine pH, saliva pH, stomach pH, blood pH, and interstitial fluids pH with a complete chemistry are all non-invasive medical tests unique to our 75 years of combined research to discover the cause of ALL sickness and disease, including ones which are idiopathic. The non-invasive medical diagnostics for ITL is called the 3D Full Body Functionality and Interstitial Fluid Testing (FBBES). [27]
New Methods for Whole Body Non-Invasive Diagnostic Testing of the Blood, Interstitial Fluids and Intracellular Fluids
Using 3-D Full Body Bio-Electro Scanning (FBBES), Full Body Thermography (FBT), Full Body Ultrasound (FBU), Live Blood Analysis (LBA) and Dried Blood Analysis (DBA) to Determine the Best Possible Strategy for Preventing and/or Reversing Any Sickness or Disease Condition and to Monitor Effective Treatment Progress [28].
In modern day conventional medicine, surgeons biopsy the lymph nodes and tissue to determine how brain disease, including malignancy is spreading to provide staging [29]. Lymphocytes, a type of white blood cell that is found in these lymph nodes which are catch-basins for acidic waste and malignant cells are responsible for breaking-down and removing cellular acidic waste and genetically mutated cells. Impaired lymphocytes and lymph nodes are at least one major factor in the many areas we test for (as described below) physiologically using functionality testing (FBBES) (as described below). The lymphatic system, the lymph nodes and the lymphocytes themselves must be functional in order to prevent and reverse any disease condition, including Interstitial Brain Disease (IBD).
Using electrodes attached to the head, hands, and feet, the functionality of the lymphatic system, circulatory system, muscular system, skeletal system, endocrine system, neurological system, reproductive system, vascular system, digestive system, and respiratory system can be non-invasively analyzed.
Interstitial chemistry, interstitial pH to determine decompensated acidosis, and the electro-conductivity of the cells to determine the state of health of all organs, glands, and tissues in the prevention and reversal of any disease condition, including IBD, associated with impaired cognitive function, diabetes mellitus, dementia and Alzheimer’s.
This FBBES also accounts for nutritional deficiencies and metabolic alkalosis or acidosis by measuring the interstitial chemistry, interstitial pH, and the electro-conductivity if the cells of the body. Measuring the pH of the interstitial fluids is more revealing as it pertains to the pH and chemistry than measuring the blood fluids for IBD condition since the blood is always trying to maintain its delicate alkaline pH of 7.365 and will not vary much. Based upon our research we have determined that IBD and all of its many symptoms are expressed as a compromised acidic environment of the interstitial fluids which may negatively affect the state of health of all body cells which make up the organs, glands and tissues, including the lung cells. [29].
It is significantly more important to measure interstitial and intracellular fluids than blood fluids in order to obtain a correct chemistry and pH when making nutritional recommendations in the prevention and treatment of IBD and any malignancy [30].
The following are quantitative measurements in healthy patients, without IBD or impaired cognitive function, dementia, Alzheimer’s or brain malignancy, comparing blood fluids with intracellular and interstitial fluids of the body compartments as a benchmark to determine deficiencies in alkalizing minerals, protein, and whether or not the patient is in decompensated acidosis of the interstitial fluids, a pre-malignancy, or malignant condition (Note: all IBD and cancer patients are in decompensated interstitial acidosis, low in interstitial sodium and high in interstitial calcium and potassium): [30]
1) Sodium: Na+ mEq/l Venous blood: 130, Arterial blood: 137, Capillary blood: 135, Intracellular fluid: 10 and Interstitial fluid: 135
2) Potassium: K+ mEq/l Venous blood: 3.2, Arterial blood: 3.5, Capillary blood: 4, Intracellular fluid: 140 and Interstitial fluid: 3.17
3) Calcium: Ca++ mEq/l Venous blood: 2.5, Arterial blood: 2.2, Capillary blood: 2.3, Intracellular fluid: 0.0001 and Interstitial fluid: 1.55
4) Magnesium: Mg mEq/l Venous blood: 0.64, Arterial blood: 0.62, Capillary blood: 0.60, Intracellular fluid: 58 and Interstitial fluid: 0.50
5) Chloride: Cl- mEq/l Venous blood: 104, Arterial blood: 101, Capillary blood: 103, Intracellular fluid: 4 and Interstitial fluid: 106
6) Bicarbonate: HCO3 mEq/l Venous blood: 22, Arterial blood: 24, Capillary blood: 23, Intracellular fluid: 10 and Interstitial fluid: 24
7) Phosphorus: P mE/l Venous blood: 2.5, Arterial blood: 2.3, Capillary blood: 2, Intracellular fluid: 75 and Interstitial fluid: 0.70
8) Sulfate: SO4 mEq/l Venous blood: 0.8, Arterial blood: 0.6, Capillary blood: 0.5, Intracellular fluid: 2 and Interstitial fluid: 0
9) Glycemia mg/dl Venous blood: 1, Arterial blood: 1, Capillary blood: 1.01, Intracellular fluid: 0.20 and Interstitial fluid: 0.90
10) Cholesterol mg/dl Venous blood: 0.66, Arterial blood: 0.630, Capillary blood: 0.676, Intracellular fluid: 0.2 and Interstitial fluid: 0.188
11) Partial Pressure of Oxygen or PO2 mmHg Venous blood: 80, Arterial blood: 90, Capillary blood: 89, Intracellular fluid: 20 and Interstitial fluid: 87.2
12) Carbon Dioxide Or PCO2 Venous blood: 46, Arterial blood: 40, Capillary blood: 42, Intracellular fluid: 50 and Interstitial fluid: 46
13) pH or potential of hydrogen Venous blood: 7.36, Arterial blood: 7.4, Capillary blood: 7.38, Intracellular fluid: 7.2 and Interstitial fluid: 7.36
14) Protein g/dl Venous blood: 72, Arterial blood: 74, Capillary blood: 73.7, Intracellular fluid: 68 and interstitial fluid: 20.6
The following are quantitative measurements in healthy patients, without IBD, impaired brain function or malignancy of the , comparing blood fluids with intracellular and interstitial fluids of the body compartments as a benchmark to determine deficiencies in alkalizing minerals, protein, and whether or not the patient is in decompensated acidosis of the interstitial fluids, a pre-malignancy, or malignant condition (Note: all IBD, impaired brain function and cancer patients are in decompensated interstitial acidosis, low in interstitial sodium and high in interstitial calcium and potassium): [30]
Results
Using non-invasive 3D-FBBES for determining the chemistry of the interstitial fluids of the whole body, including the brain is not only a test for determining decompensated acidosis or compensated alkalosis (a pre-condition of IFBD) of the ALL interstitial fluids of the body, including the brain, but is an effective way to determine efficacy of any medical or natural alkalizing therapy in prevention and treatment of ANY sickness or disease. The results of the using 3D-GBBES combined with conventional medical blood tests and/or the Non-Invasive Blood Tests [NIBT] covering over 170 parameters of the blood, including blood counts, chemistry, pH of the blood, stomach and most important quantifying acid loads, such as lactic acid with a 98 percent accuracy for determining pre-interstitial-fluid brain disease and IFBD and its many symptomologies as noted above.
References
1. Bacyinski, A; Xu, M; Wang, W; Hu, J (2017). “The Paravascular Pathway for Brain Waste Clearance: Current Understanding, Significance and Controversy”. Frontiers in Neuroanatomy. 11: 101. doi:10.3389/fnana.2017.00101. PMC 5681909. PMID 29163074.
2. Kiviniemi, Vesa J; et al. (2016). “Ultra-fast magnetic resonance encephalography of physiological brain activity – Glymphatic pulsation mechanisms?”. J Cereb Blood Flow Metab. 36 (6): 1033–1045. doi:10.1177/0271678X15622047. PMC 4908626. PMID 26690495.
3. Konnikova, Maria (11 January 2014). “Goodnight. Sleep Clean”. The New York Times. Retrieved 18 February2014. She called it the glymphatic system, a nod to its dependence on glial cells
4. Jump up to:a b Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD, Derecki NC, Castle D, Mandell JW, Lee KS, Harris TH, Kipnis J (July 2015). “Structural and functional features of central nervous system lymphatic vessels”. Nature. 523 (7560): 337–41. doi:10.1038/nature14432. PMC 4506234. PMID 26030524.
5. Jump up to:a b Aspelund A, Antila S, Proulx ST, Karlsen TV, Karaman S, Detmar M, Wiig H, Alitalo K (June 2015). “A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules”. The Journal of Experimental Medicine. 212 (7): 991–9. doi:10.1084/jem.20142290. PMC 4493418. PMID 26077718.
6. Bert JL; Pearce RH (1984). The interstitium and microvascular exchange. In: Handbook of Physiology. The Cardiovascular System. Microcirculation (sect. 2; pt. 1; chapt. 12; vol. IV ed.). Bethesda, MD: American Physiological Society. pp. 521–547. ISBN 0683072021.
7. Wiig, H; Swartz, M. A (2012). “Interstitial fluid and lymph formation and transport: Physiological regulation and roles in inflammation and cancer”. Physiological Reviews. 92 (3): 1005–60. doi:10.1152/physrev.00037.2011. PMID 22811424.
8. Scallan J; Huxley VH; Korthuis RJ (2010). The Interstitium. In: Capillary Fluid Exchange: Regulation, Functions, and Pathology. San Rafael, CA: Morgan & Claypool Life Sciences.
9. Young, RO, Migalko, G (2019). Are the Interstitial Fluids Raining Acid on YOUR Lung Cells? Interstitial Fluid Lung Disease (IFLD) of the Interstitium Organ The Cause and Self-Care to a Self-Cure for Lung Disease. International Journal of Cancer Research and Therapy, January, 2020.
510. Mathieu E, Gupta N, Macdonald RL, Ai J, Yücel YH. In vivo imaging of lymphatic drainage of cerebrospinal fluid in mouse. Fluids Barriers CNS. 2013;10:35. [PMC free article] [PubMed] [Google Scholar]
11. Cserr HF, Harling-Berg CJ, Knopf PM. Drainage of brain extracellular fluid into blood and deep cervical lymph and its immunological significance. Brain Pathol Zurich Switz. 1992;2:269–276. [PubMed] [Google Scholar]
12. Mehler MF, Gokhan S (2000). “Mechanisms underlying neural cell death in neurodegenerative diseases: alterations of a developmentally-mediated cellular rheostat”. Trends Neurosci. 23 (12): 599–605. doi:10.1016/s0166-2236(00)01705-7. PMID 11137149.
13. Narasimhan K (March 2006). “Quantifying motor neuron loss in ALS”. Nature Neuroscience. 9 (3): 304. doi:10.1038/nn0306-304. PMID 16498424.
14. Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA, Nagelhus EA, Nedergaard M (August 2012). “A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β”. Science Translational Medicine. 4(147): 147ra111. doi:10.1126/scitranslmed.3003748. PMC 3551275. PMID 22896675.
15. Gauberti, Gaberel (2014). “Impaired Glymphatic Perfusion After Strokes Revealed by Contrast-Enhanced MRI: A New Target for Fibrinolysis?”. Stroke.[permanent dead link]
16. Harris MG, et al. Immune privilege of the CNS is not the consequence of limited antigen sampling. Sci Rep. 2014;4:4422. [PMC free article] [PubMed] [Google Scholar]
17. Laman JD, Weller RO. Drainage of cells and soluble antigen from the CNS to regional lymph nodes. J Neuroimmune Pharmacol Off J Soc NeuroImmune Pharmacol. 2013;8:840–856. [PubMed] [Google Scholar]
18. Schneider M, Ny A, Ruiz de Almodovar C, Carmeliet P. A new mouse model to study acquired lymphedema. PLoS Med. 2006;3:e264. [PMC free article] [PubMed] [Google Scholar]
19. Kida S, Pantazis A, Weller RO. CSF drains directly from the subarachnoid space into nasal lymphatics in the rat. Anatomy, histology and immunological significance. Neuropathol Appl Neurobiol. 1993;19:480–488. [PubMed] [Google Scholar]
20. Xie L, et al. Sleep drives metabolite clearance from the adult brain. Science. 2013;342:373–377. [PMC free article] [PubMed] [Google Scholar]
21. Yang L, et al. Evaluating glymphatic pathway function utilizing clinically relevant intrathecal infusion of CSF tracer. J Transl Med. 2013;11:107. [PMC free article] [PubMed] [Google Scholar]
22. . Berton M, et al. Generalized lymphedema associated with neurologic signs (GLANS) syndrome: A new entity? J Am Acad Dermatol. 2015;72:333–339. [PubMed] [Google Scholar]
23. Akiyama H, et al. Inflammation and Alzheimer’s disease. Neurobiol Aging. 2000;21:383–421. [PMC free article] [PubMed] [Google Scholar]
24. Hohlfeld R, Wekerle H. Immunological update on multiple sclerosis. Curr Opin Neurol. 2001;14:299–304. [PubMed] [Google Scholar]
25. Abbott NJ (2004). “Evidence for bulk flow of brain interstitial fluid: significance for physiology and pathology”. Neurochem. Int. 45 (4): 545–52. doi:10.1016/j.neuint.2003.11.006. PMID 15186921.
26. Ryu JH, Olson EJ, Midthun DE, Swensen SJ (November 2002). “Diagnostic approach to the patient with diffuse lung disease”. Mayo Clinic Proceedings. 77 (11): 1221–7, quiz 1227. doi:10.4065/77.11.1221. PMID 12440558.
27. Galina Migalko, Universal Medical Imaging Group, Valley Village, California, USA. www.universalmedicalimaging.com
28. Ronald G.CrystalM.D., James E.GadekM.D., Victor J.FerransM.D., Jack D.FulmerM.D., Bruce R.LineM.D., Gary W.HunninghakeM.D, “Review Interstitial lung disease: Current concepts of pathogenesis, staging and therapy”. The American Journal of Medicine
Volume 70, Issue 3, March 1981, Pages 542-568.
29. Rose BD (2001) Post TW. Clinical Physiology of Acid-Base and Electrolyte Disorders, (5th edn), McGraw-Hill, New York. p. 347.
30. Young RO, Migalko G (2015) Alkalizing Nutritional Therapy in the Prevention and Reversal of any Cancerous Condition. Int J Complement Alt Med 2(1): 00046. DOI: 10.15406/ijcam.2015.02.00046.



